Is bleach made from salt?
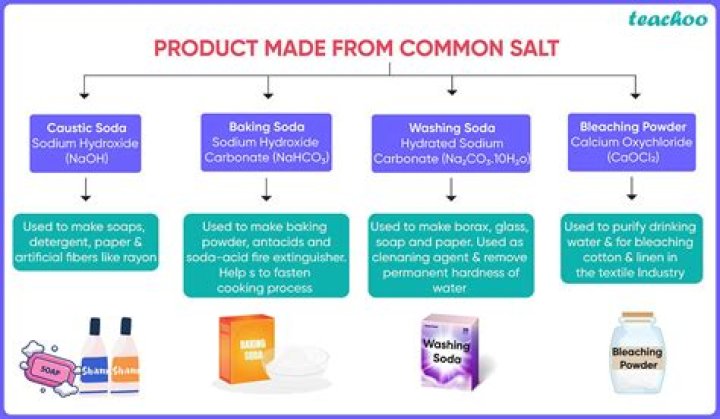
Answer. The active ingredient in household bleach is sodium hypochlorite, which is derived from salt. In fact, Clorox is headquartered in Oakland, California because we started making bleach here by running electricity through salt water from San Francisco Bay! So household bleach begins and ends as salt and water.
What is bleach made of?
What is bleach? Household bleach is actually a mixture of chemicals, Its main constituent is a solution of ~3-6% sodium hypochlorite (NaOCl), which is mixed with small amounts of sodium hydroxide, hydrogen peroxide, and calcium hypochlorite.Is bleach a sodium salt?
Which brings us to what is arguably the most widely-used bleaching compound in the world, chlorine dioxide, its chemical symbol is Cl-O2, a combination of chlorine and oxygen. It is made from a salt called sodium chlorite, Na-Cl-O2.Does bleach convert salt?
When you buy a bottle of chlorine bleach at the store, what you are buying is sodium hypochlorite mixed with water. Sodium hypochlorite is NaOCl. Since table salt is NaCl, bleach is very similar to table salt and reverts to table salt if left out.Is bleach a base or salt?
Conclusion. Bleach or sodium hypochlorite is a base and its pH ranges between 11 and 13. In an aqueous solution, it dissociates to form sodium and hypochlorite ions. The ClO- ions further react with water molecules resulting in the formation of hydroxide ions.Turning Ocean Water into Chlorine Bleach | How To Make Bleach, Hydrogen Gas, or Smurf Juice
What is Clorox bleach made of?
The ingredients in Clorox bleach are water, sodium hypochlorite, sodium chloride, sodium carbonate, sodium chlorate, sodium hydroxide and sodium polyacrylate.Is bleach same as chlorine?
Chlorine is a natural element and an ingredient of bleach, while bleach is a solution and the product of combining chlorine and other chemicals. 2. Chlorine exists in nature, while bleach is a manufactured product.What is bleach chemical?
Bleach is a chemical that is diluted and sold for household use. It is a mixture of water and the chemical sodium hypochlorite. For uses in the home and many workplaces, it is usually sold with concentrations of sodium hypochlorite present at a range from about 3 to 9 percent.Can you mix table salt and bleach?
When dissolved in water, bleach provides OH-. There will be a neutralization reaction if the salt you use is acidic. There could be a reaction if the salt you use is basic. The salt will be dissolved in a bleach solution.Is sodium hypochlorite the same as salt?
Sodium chlorite and sodium hypochlorite are common sodium salts used in industries. Both these compounds are very important as bleaching agents and disinfectants.What is bleach formula?
It is usually called bleach, because it is the active ingredient in bleach. The chemical formula is NaClO and consists of one atom of sodium (Na), one atom of chlorine (Cl) and one atom of oxygen (O).Does bleach turn into sodium chloride?
Bleach can react violently with hydrogen peroxide and produce oxygen gas: H2O2 (aq) + NaOCl (aq) → NaCl (aq) + H2O (aq) + O2 (g)How do you make bleach from scratch?
To mix chlorine bleach combine 5.25 percent sodium hypochlorite and 94.75 percent water. To mix non-chlorine bleach, combine equal parts hydrogen peroxide and water. Pour mixture into the plastic container, and secure tightly. Shake to mix ingredients.How do you make homemade bleach?
Homemade bleach alternative recipe
- ½ cup 3% hydrogen peroxide.
- 2 tablespoons lemon juice OR ½ teaspoon citric acid.
- 3 ¼ cups water.
- 5 drops lemon essential oil.
Is bleach found in nature?
Bleach is from the organochlorine family of chemicals, compounds rarely found in nature and which can take centuries to decompose.Is hydrogen peroxide a bleach?
Hydrogen peroxide is an oxygen-based, non-chlorine bleach. Although not as powerful as chlorine bleach, it is safer for both humans and the environment because it breaks down into water and oxygen.What pH is bleach?
Bleach: pH 11-13Bleach is one of the most common cleaning supplies in households and commercial settings. This particular product has a pH between 11 and 13. Its high level of alkalinity is what makes it corrosive. As a result, ventilation is important when using bleach.



